Product Component
The product formulation includes the following components:
Cross-linked Sodium Hyaluronate (20 mg/mL, crosslinked with BDDE)
Uncross-linked Sodium Hyaluronate
Lidocaine Hydrochloride (3 mg/mL)
Sodium Chloride
Phosphate Buffer System
Water for Injection (WFI)
Additional Information
Sodium hyaluronate is of non-animal origin, produced by microbial fermentation.
The gel particles are encapsulated in a pre-filled sterile glass syringe.
The product includes two 30G SUS304 stainless steel injection needles.
Sterilization methods:
Gel suspension: Moist heat sterilization
Injection needles: Ethylene oxide sterilization
Shelf life: 36 months (single-use product)

Core Technological Advantages
●Dual-phase hyaluronic acid technology
The patented cross-linking HA structure allows the ingredients to be evenly dispersed beneath the skin, delivering both an instant volumizing result and a sustained boost in collagen production.
●Professional-level injection experience
Equipped with 30G ultra-fine needles, it reduces injection trauma. Combined with the pain-relieving ingredient lidocaine, it significantly enhances the comfort of treatment.
●International certification guarantee
It has obtained ISO13485 and CE international certifications. The manufacturing environment complies with GMP Class 100 cleanroom standards, and quality control is maintained according to the Six Sigma benchmark.
Treatment Area
Hydrofill 2ML Facial Filler is designed based on the mechanism of skin aging and provides customized solutions for the aging characteristics of different parts. Its differentiated injection technology can precisely meet the youthful demands of multiple areas on the face and body.
Injection Site | Effect |
|---|---|
Facial rejuvenation | Precise treatment by zones |
Middle and lower face | Effectively improve the depression of nasolabial folds and the shadow of tear troughs, and reshape a soft contour. Lip area: Fade marionette lines, enhance the hydration of the lips, and improve dryness and flaking. |
Forehead and jaw | Optimize the forehead line and tighten the jaw contour through deep dermal injection. |
Overall youthfulness | Harmonious aesthetic shaping |
Neck and hands | Deeply fill the neck creases, restore the volume of the back of the hand and hide the veins |
Exposed areas | Repair skin quality issues caused by light damage in areas such as the chest, elbows, and knees |
Creased areas | Increase the moisture content of the skin in areas such as the armpits and enhance the surface smoothness |
The treatment plan should be tailored to individual differences. Professional physicians should select the injection strategy for the middle dermis (fine line repair/moisturizing) or deep dermis (contour lifting/volume replenishment) based on the thickness of the skin and the characteristics of muscle activity.
Cross-linked Hyaluronic Acid Sodium Gel
Key attributes
Ingredients | 20mg/ml Cross-linked sodium hyaluronate |
|---|---|
Gel Hardness | Soft |
Lifting Power | N / A |
Usage | Improve skin appearance |
Injection Levels | Deep dermis |
Product Size | 1mL(One syringe, two needles) |
Suggested Needles | 30 G Sharp needles |
Suggested Treatment Plan | 3 injections, 4 weeks apart |
FAQs
1. Why does cross-linked hyaluronic acid last longer than non-crosslinked HA?
Cross-linked hyaluronic acid has a stable three-dimensional structure that resists natural enzymatic degradation in the skin. While non-crosslinked HA breaks down within a few days to a week, cross-linked HA remains in the body for several months, providing longer-lasting filling and moisturizing effects. Our factory uses a high-efficiency BDDE cross-linking process to ensure stability and consistent quality for medical aesthetic applications.
2. Is your cross-linked hyaluronic acid derived from animals?
No. Our sodium hyaluronate is produced through microbial fermentation, ensuring it is non-animal origin and biocompatible. This minimizes the risk of allergic reactions and ensures high purity for use in dermal fillers, mesotherapy, and regenerative formulations.
3. What is the purpose of lidocaine in the injectable gel?
Lidocaine (0.3%) is included to reduce discomfort during injection. It provides local pain relief and enhances the treatment experience for patients. Each batch of our product is strictly quality-controlled to ensure consistent lidocaine concentration and safe performance in clinical use.
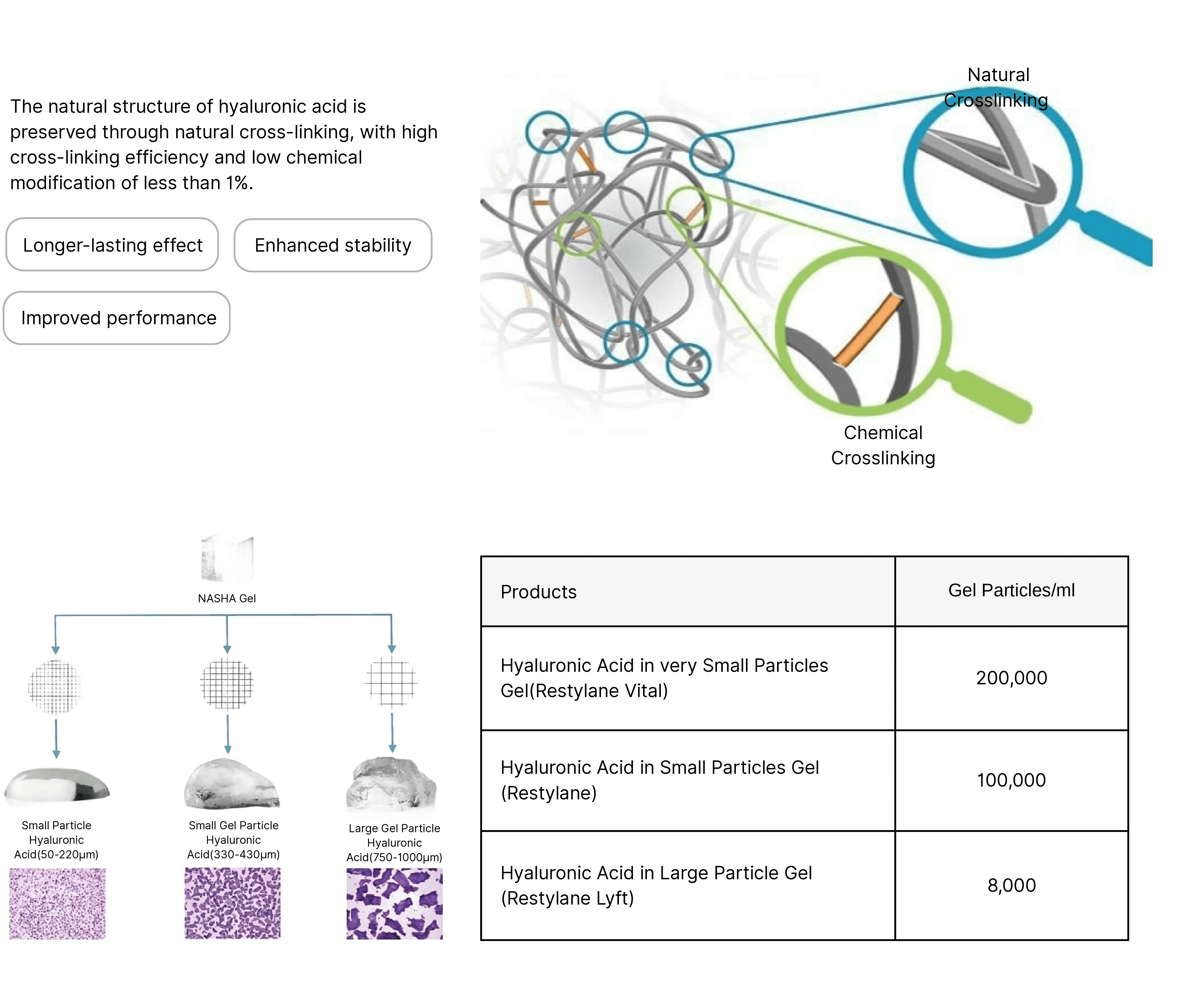
4. How does your cross-linking process preserve the natural structure of hyaluronic acid?
Our process uses low-level BDDE cross-linking (less than 1% chemical modification) to stabilize the hyaluronic acid molecules while maintaining their natural biological structure. This balance results in excellent elasticity, biocompatibility, and hydration retention—ideal for facial contouring and anti-aging treatments.
5. How can clinics or distributors work with your factory for OEM or bulk supply?
We are a professional manufacturer and global supplier specializing in medical-grade cross-linked hyaluronic acid. Clinics, brands, and distributors can collaborate with us for OEM, private labeling, or bulk supply. We provide full technical documentation, quality certifications, and global logistics support to ensure safe and efficient delivery.












