Type
Contour
Application
Suitable for individuals aged 18 and above to correct facial wrinkles and deep skin depressions, restore cheek fullness, address marionette lines, and sculpt the jawline contour.
Place of Origin
China
Brand Name
Rveiling®
PLLA-PEG Injectable Facial Filler Overview
Rveiling® PLLA-PEG Injectable Facial Filler is a milky white, sterile, biodegradable viscous gel implant. Utilizing a pre-filled syringe system, it combines instant filling with long-lasting effects to restore skin tissue volume through injection, achieving facial rejuvenation and aesthetic contouring.
1.Vial Details
Ingredients: Sodium Hyaluronate, PLLA-PEG Microspheres, Lidocaine Hydrochloride, BDDE Crosslinking Agent, Sodium Chloride, Injection Water Phosphate Buffer System.
Stability: 24-month shelf life through irradiation sterilization.
Specifications: 1 mL/syringe, 2 mL/syringe, 3 mL/syringe
Adjustable concentrations support versatile applications across cheeks, temples, forehead, and hands.
2.Structural Composition
Rveiling® PLLA-PEG Injectable Facial Filler is a composite gel system. Its primary components include: PLLA-PEG microspheres, active ingredients at a formulation concentration of 20.7%, responsible for long-term stimulation of collagen regeneration. Cross-linked sodium hyaluronate gel: suspension carrier, providing immediate filling support. Sodium hyaluronate is labeled at 16mg/mL, produced via microbial fermentation and crosslinked with BDDE. The gel carrier additionally contains 0.3% lidocaine hydrochloride to effectively reduce discomfort during injection and enhance treatment comfort. All components are sealed within a single-use, pre-filled syringe.
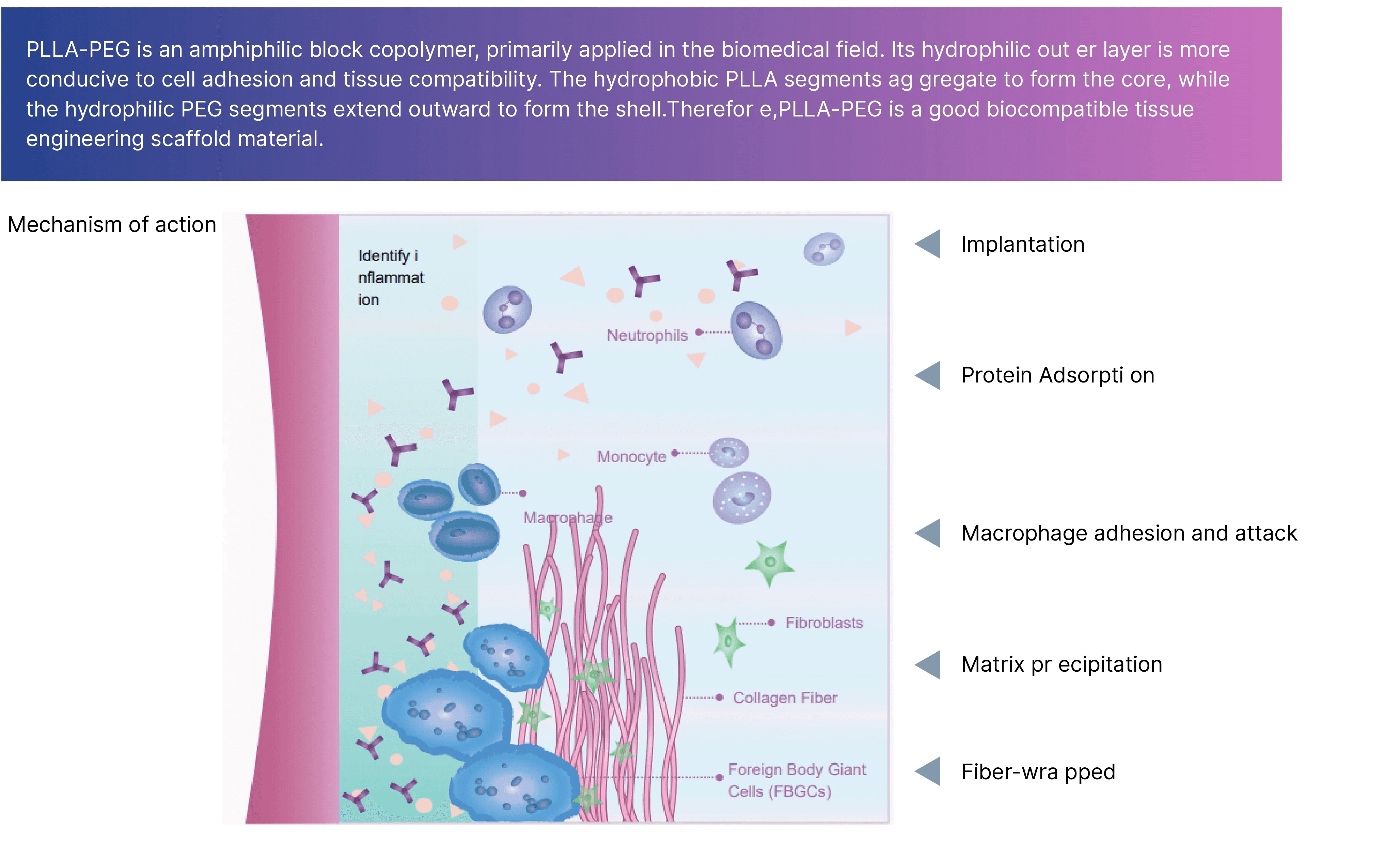
Mechanism of Action
Following injection, the PLLA-PEG facial filler is implanted into the deep dermis and superficial periosteum for physical augmentation. Immediate improvement in skin volume and contour is achieved through the action of sodium hyaluronate gel particles.
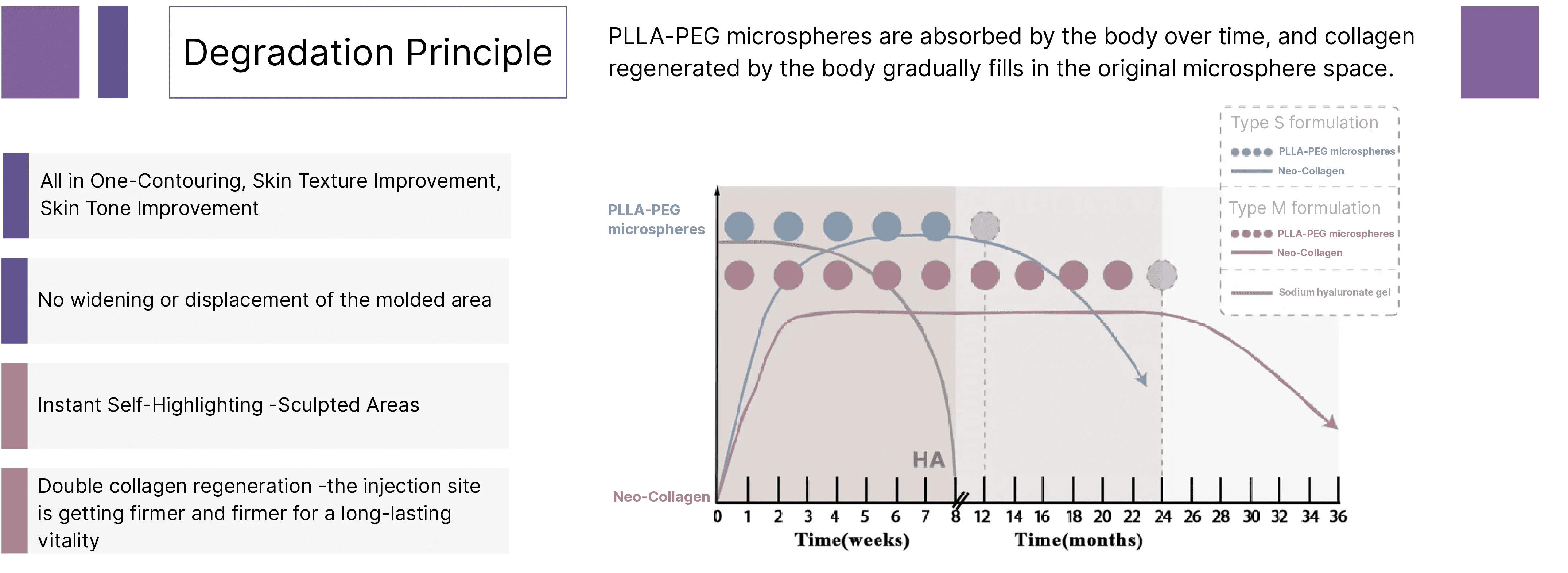
Rveiling® Injectable PLLA-PEG Composite Facial Filler achieves long-lasting collagen regeneration through a “guided regeneration” mechanism. Its action begins with a temporary scaffold formed by degradable PLLA-PEG microspheres and gel. Within weeks of implantation, the material initiates controlled degradation. This process is recognized by the body as a mild regenerative signal rather than an inflammatory response, precisely recruiting reparative macrophages and fibroblasts. Within the locally formed regenerative microenvironment, fibroblasts are activated and synthesize large quantities of Type I and Type III collagen. Newly synthesized collagen deposits spatially along the original scaffold, while the PLLA material fully degrades over subsequent months. This achieves a natural transition from “physical filling” to “biological regeneration,” enabling long-term, natural volume maintenance and rejuvenation effects through newly formed autologous collagen tissue. PLLA-PEG microspheres gradually degrade through ester bond hydrolysis into lactic acid, which is ultimately metabolized into carbon dioxide and water and excreted through natural physiological processes.
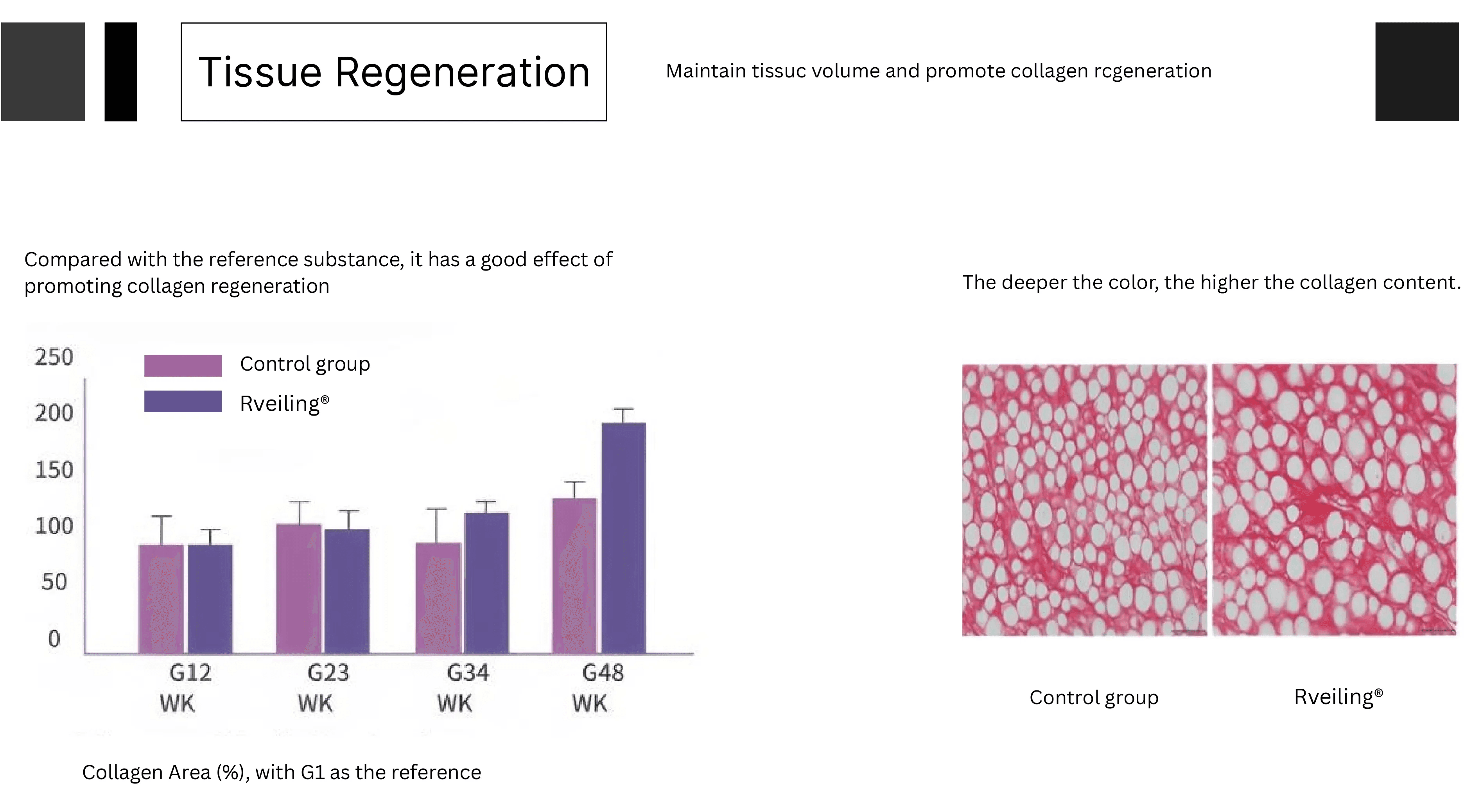
Long-lasting Maintenance
The PLLA-PEG microspheres contained in the product act as a biological scaffold, continuously promoting the regeneration of endogenous collagen. This extends and optimizes the filling effect, delivering natural, enduring tissue support and skin texture improvement.
1. Patient Suitability
Ideal for anti-aging seekers desiring firmness and contour enhancement.
Requires physician review for allergies, keloids, pregnancy, or breastfeeding.
Core Technology Edge
The hyaluronate carrier system ensures even microsphere distribution, exact dosing from pre-filled vials, and inflammation reduction via high biocompatibility. This innovation powers safer, more predictable PLLA-PEG injectable filler outcomes for clinical excellence.
B2B Factory Solutions
Collaborate with our Poly-L-Lactic Acid-Polyethylene Glycol facility for bulk PLLA-PEG microspheres production, custom formulations, and white-label packaging. Access competitive pricing, rapid scaling, and global supply chain support to dominate the facial implant filler market.
Additional Information
1.Indications
This product is indicated as a dermal filler for injection into the deep dermis, deep subcutaneous tissue, and superficial periosteum to correct facial depressions and contour laxity caused by tissue volume loss.
2.Sterilization Method
The gel in the pre-filled syringe is sterilized by high-temperature steam; the accompanying injection needles are sterilized by ethylene oxide. Usage Guidelines: This product is for single-use only.
3.Precautions
Strict adherence to sterile surgical procedures is mandatory during use. Refer to the enclosed instructions for detailed usage guidelines.
Rveiling® PLLA-PEG filler microspheres continuously stimulate fibroblasts to induce the secretion and synthesis of collagen in the human body. Consequently, they are widely used in medical aesthetic fillers and tissue regeneration and repair therapies.
Rveiling® PLLA-PEG Microspheres Continuously Induce Human Fibroblasts to Secret and Synthesize Collagen

Rveiling® PLLA-PEG Microspheres Appearance: Smooth spherical PLLA-PEG microsphere particles, non-toxic, extremely safe, non-irritating, uniform particle size, and comparative biological response effects upon implantation into soft tissue.
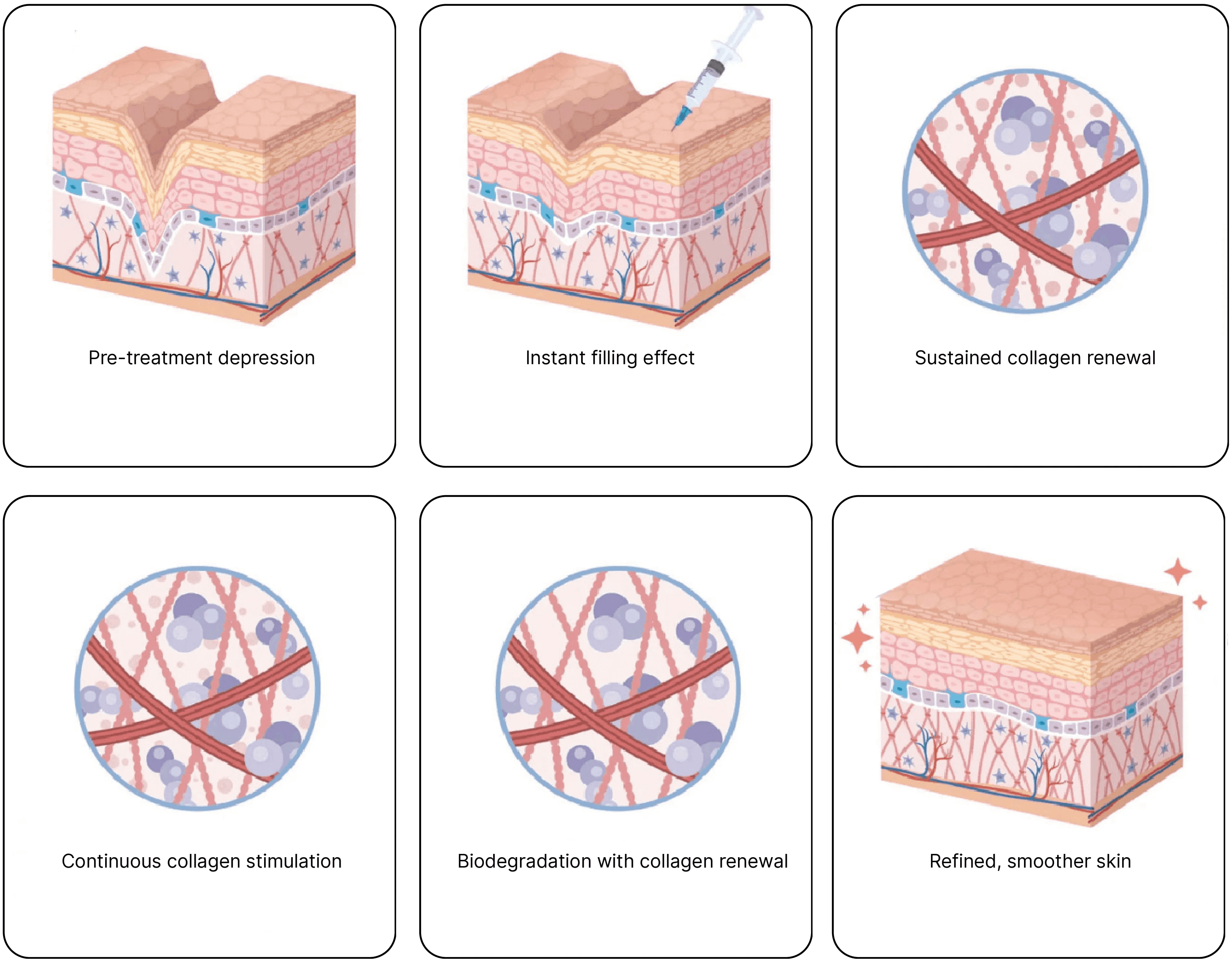
Rveiling® PLLA-PEG microspheres undergo gradual degradation within tissue, stimulating the progressive regeneration of endogenous collagen. As new collagen replaces the original space occupied by the microspheres, it achieves natural filling and structural support effects.
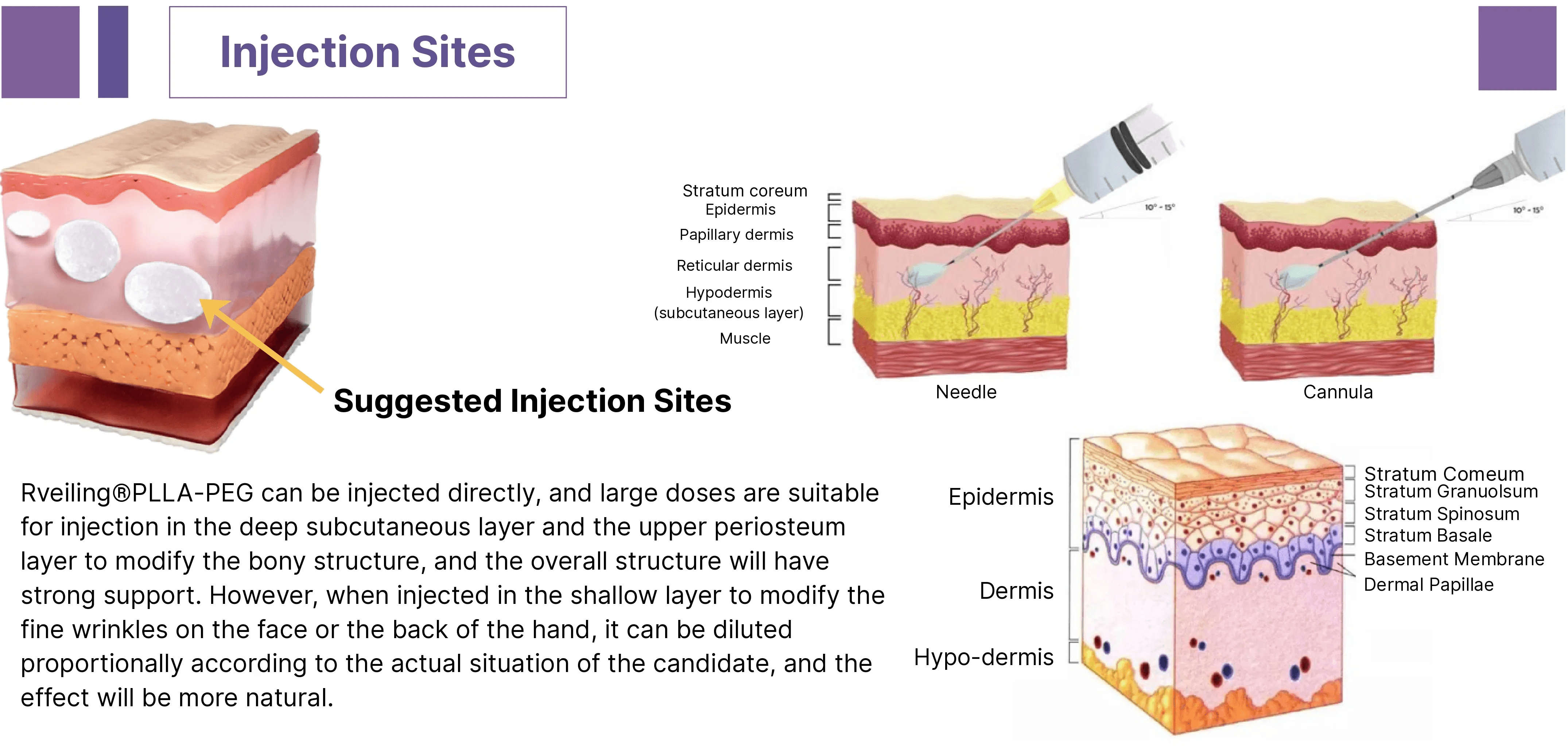
Rveiling® PLLA-PEG Facial Filler delivers deep structural support or natural, three-dimensional contouring effects depending on the injection layer.
Poly-L-Lactic Acid-Polyethylene Glycol Product Status

Key attributes
Specification | 1 mL/syringe, 2 mL/syringe, 3 mL/syringe |
|---|---|
Storage condition | 2-10℃, prohibit freezing, avoid sunlight. |
Transport conditions | Transported at normal temperature, the transport cycle should not exceed1 month. |
Usage Restriction | Do not re-use |
Sterilization Method | High-temperature steam sterilization |
Usage Warning | Do not use if packaging is damaged |
Sterilization Restriction | Do not resterilize |
Cross-linked Sodium Hyaluronate | 16 mg/mL |
Poly(L-lactic acid)-poly(ethylene glycol) microspheres | 20.7 % |
Lidocaine Hydrochloride | 3 mg/mL |
FAQs
1.What is Rveiling® PLLA-PEG Injectable Facial Filler ?
Rveiling® PLLA-PEG Injectable Facial Filler utilizes PLLA-PEG material with excellent biocompatibility to produce microspheres of uniform particle size. PLLA-PEG is an amphiphilic block copolymer primarily applied in biodegradable medical fields. Its hydrophilic outer layer enhances cell adhesion and tissue compatibility. The hydrophobic PLLA segments aggregate to form the core, while the hydrophilic PEG segments extend outward to create the shell. The microspheres are suspended within a gel particle carrier. Following injection and implantation, the gel particles gradually degrade, while the microspheres provide long-term structural support and continuously induce human fibroblasts to secrete and synthesize collagen. Over time, PLLA-PEG undergoes gradual biodegradation.
2. Can outer packaging be customized for OEM or private label production?
Yes. As a professional B2B manufacturer and supplier, we offer OEM and ODM customization services, including logo and packaging design. We support clinics, distributors, and medical aesthetic brands with flexible collaboration models.
3. What are the primary applications of Rveiling® injectable PLLA-PEG facial filler?
Rveiling® PLLA-PEG injectable implant filler is an off-white, viscous, sterile, latex-free, pyrogen-free, semi-solid gel, biodegradable dermal implant filler with immediate results. This implant filler is indicated for correcting facial wrinkles and deep dermal depressions, cheek augmentation, marionette lines, jawline contouring, and restoring/correcting facial fat loss (atrophy) in AIDS patients. Injected into the soft tissue above the periosteum, it provides non-permanent yet long-lasting enhancement.
4. How long do the effects of Rveiling® PLLA-PEG facial filler last?
Typically, results persist for 12 to 24 months, depending on the injection area, the clinician's technique, and the patient's metabolism. The sodium hyaluronate gel carrier delivers immediate filling effects. The gel particles degrade before the microspheres, while the PLLA-PEG microspheres continue to stimulate human fibroblasts to secrete and synthesize collagen, providing long-lasting volume and firmness over time.
5. What certifications and quality standards do you offer for B2B partners?
Our manufacturing strictly adheres to ISO 13485, GMP, and CE certification—globally recognized standards for medical device production quality and safety. Each production batch undergoes rigorous full-performance biological testing, covering biocompatibility, sterility, and all product performance metrics. This stringent process ensures 100% batch compliance, delivering consistent reliability you can trust.
Other Products













