Type
Contour
Application
Correcting moderate to severe facial contouring, chin, cheekbone, and jawline sculpting
Place of Origin
China
Brand Name
Rveiling
1. Product Overview of Rveiling® CaHA Injectable Facial Filler
What is HA-CaHA Injectable Facial Filler?
Milky White Biodegradable Gel Implant
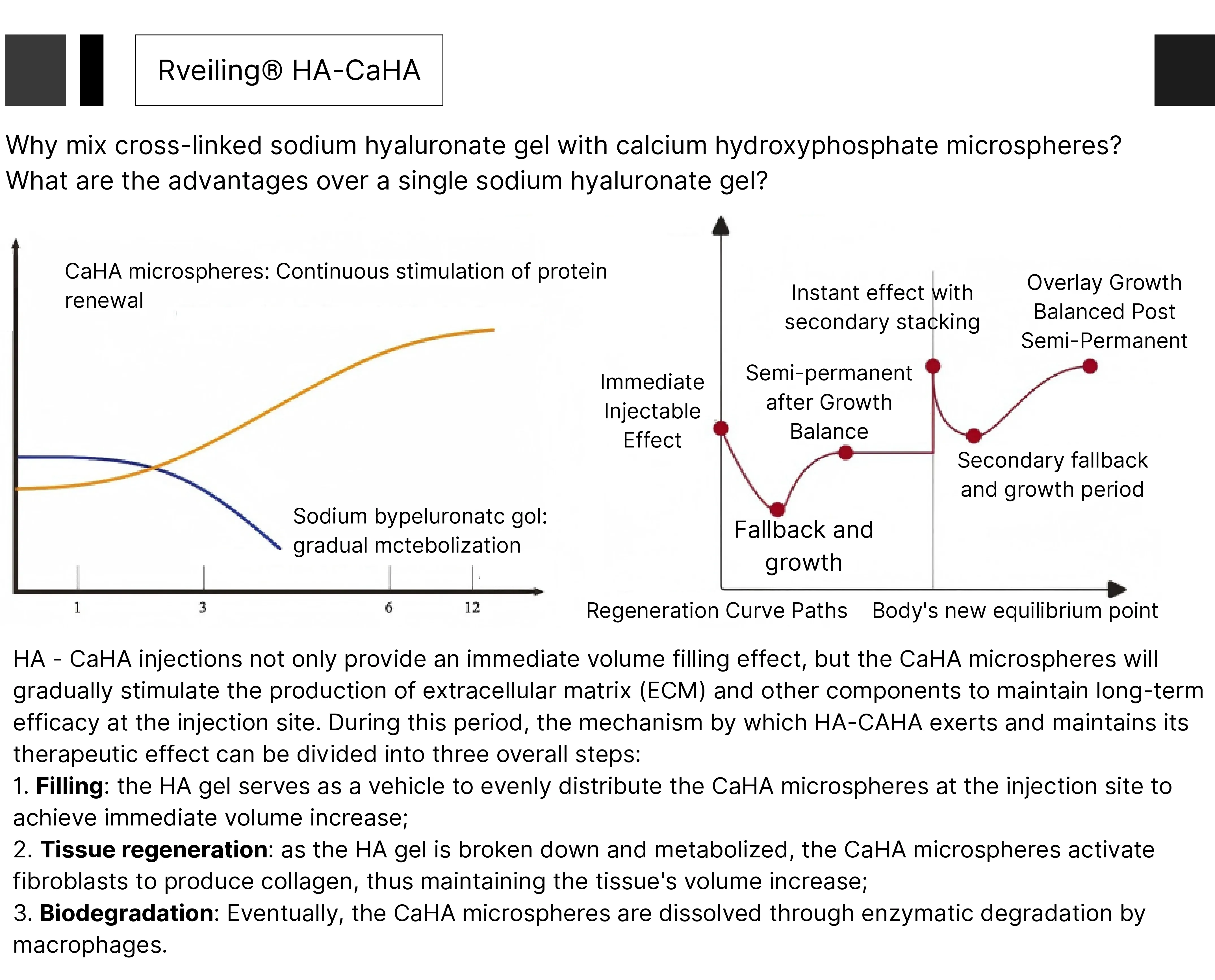
Rveiling® HA-CaHA Injectable Facial Filler is a milky white, sterile, biodegradable viscous gel implant. Utilizing a pre-filled syringe system, it combines instant filling with long-lasting volume retention. This product is indicated for injectable restoration of skin tissue volume to achieve facial rejuvenation and aesthetic contouring.
Key Benefits for Facial Rejuvenation
Product Performance
The filler combines the hydrating properties of sodium hyaluronate with calcium hydroxyapatite microspheres, achieving immediate facial volume restoration while promoting collagen synthesis for long-term rejuvenation. Ideal for cheek augmentation, wrinkle correction, and facial contouring.
2.Product Specifications
1.Syringe Volume Options
Flexible Dosing for Different Treatments
Available in 1 mL, 2 mL, and 3 mL pre-filled syringes to meet diverse clinical requirements.
2.Shelf Life
36-Month Stability
Store in a cool, dry environment. Single-use only for consistent clinical outcomes.
3.Usage Guidelines
This product is for single-use only.
3. CaHA Injectable Facial Filler Structure and Composition
Active Ingredients
High-Quality Biocompatible Components
Sodium Hyaluronate (20 mg/mL) – Provides hydration and immediate tissue filling.
Calcium Hydroxyapatite Microspheres (30.7%) – Forms a scaffold for collagen regeneration and long-lasting contour.
Lidocaine Hydrochloride (0.3%) – Reduces injection discomfort.
Sodium Chloride & Phosphate Buffer System – Maintains physiological pH and biocompatibility.
Sterile Injection Water – Ensures a stable gel consistency.
Structure and Composition
Rveiling® HA-CaHA Injectable Facial Filler is a milky white viscous gel, sterile, latex-free, pyrogen-free, semi-solid, biodegradable dermal implant. It primarily consists of synthetic calcium hydroxyapatite microspheres at a concentration of 30.7%, suspended within a cross-linked sodium hyaluronate gel carrier.
The gel carrier mainly comprises sodium hyaluronate, lidocaine hydrochloride, BDDE cross-linking agent, sodium chloride, phosphate buffer system, and water for injection. The sodium hyaluronate is produced via microbial fermentation, with a labeled concentration of 20mg/ml sodium hyaluronate and 0.3% lidocaine hydrochloride. Encapsulated in a single-use, pre-filled syringe. The addition of lidocaine hydrochloride reduces pain during treatment and enhances patient comfort.
4. Product Performance
Particle Size and Injection Precision
Safe and Effective Dermal Filler
Rveiling® HA-CaHA Injectable Facial Filler primarily consists of cross-linked sodium hyaluronate gel particles and CaHA microspheres. The median particle size of CaHA microspheres is 32μm ± 6μm, with at least 95% of microspheres measuring between 25 and 50μm. This product serves as a dermal filler, injected into the deep dermis, deep subcutaneous tissue, and superficial periosteum to enhance skin volume. It delivers immediate results at the injection site, while the CaHA microspheres prolong the filling effect.
Mechanism of Action
From Physical Filling to Biological Regeneration
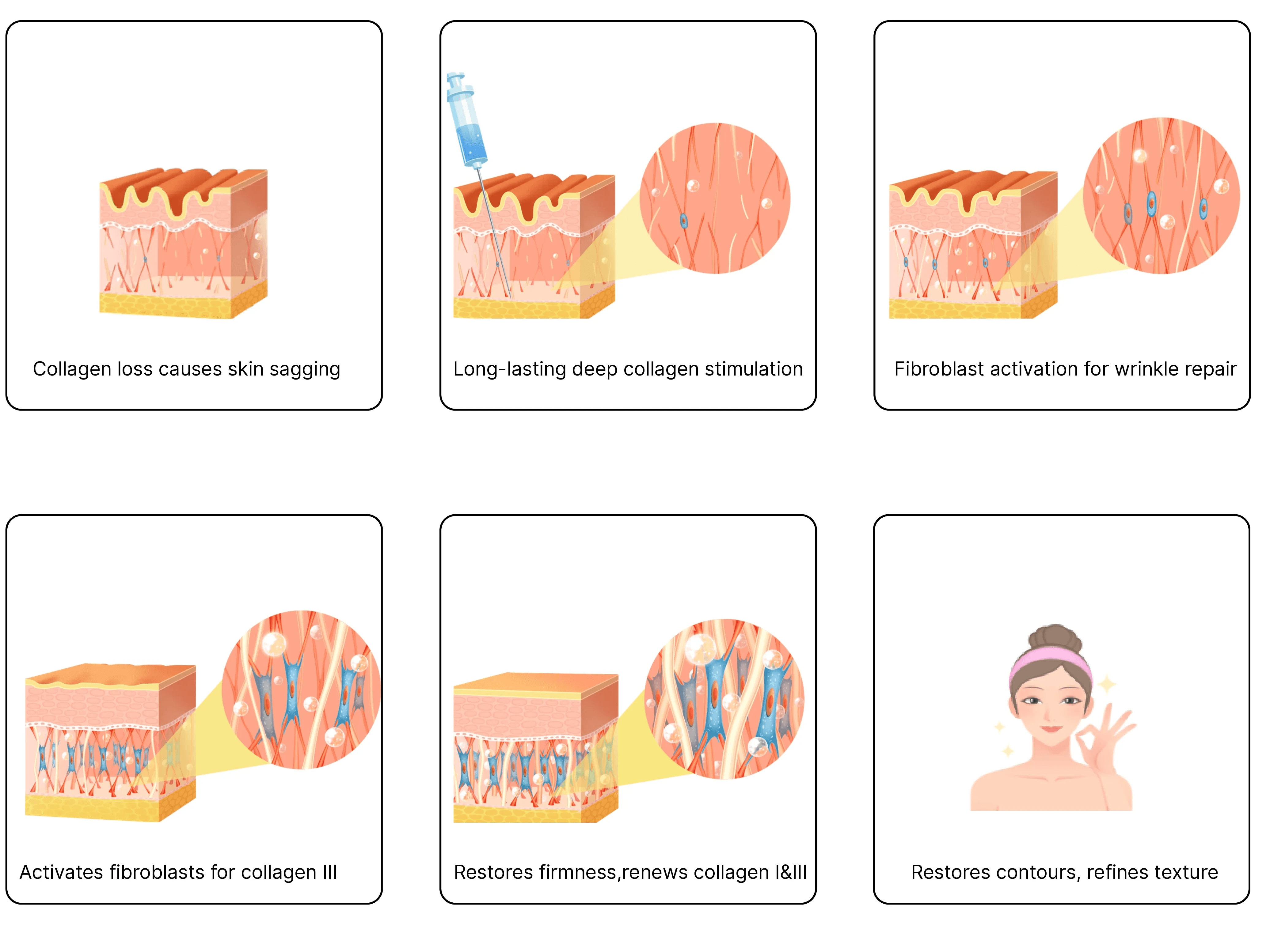
Rveiling® Calcium Hydroxyapatite (CaHA) Injectable Facial Filler achieves soft tissue augmentation and rejuvenation through a synergistic mechanism. Its action begins with a composite system: the gel carrier provides immediate volume while subsequently biodegrading; the retained CaHA microspheres form a biological scaffold that activates the body's beneficial repair response.
This process precisely recruits key reparative cells like macrophages and fibroblasts, forming a “regenerative microenvironment” around the microspheres. Through sustained biochemical and mechanical signaling, it powerfully activates fibroblast synthetic functions, efficiently secreting Type I and Type II collagen to form a novel autologous collagen network. Ultimately, the CaHA microspheres degrade slowly after guiding collagen regeneration, achieving a seamless transition from “physical filling” to “biological regeneration” in therapeutic action. This delivers natural, long-lasting rejuvenation effects.
5. Indications
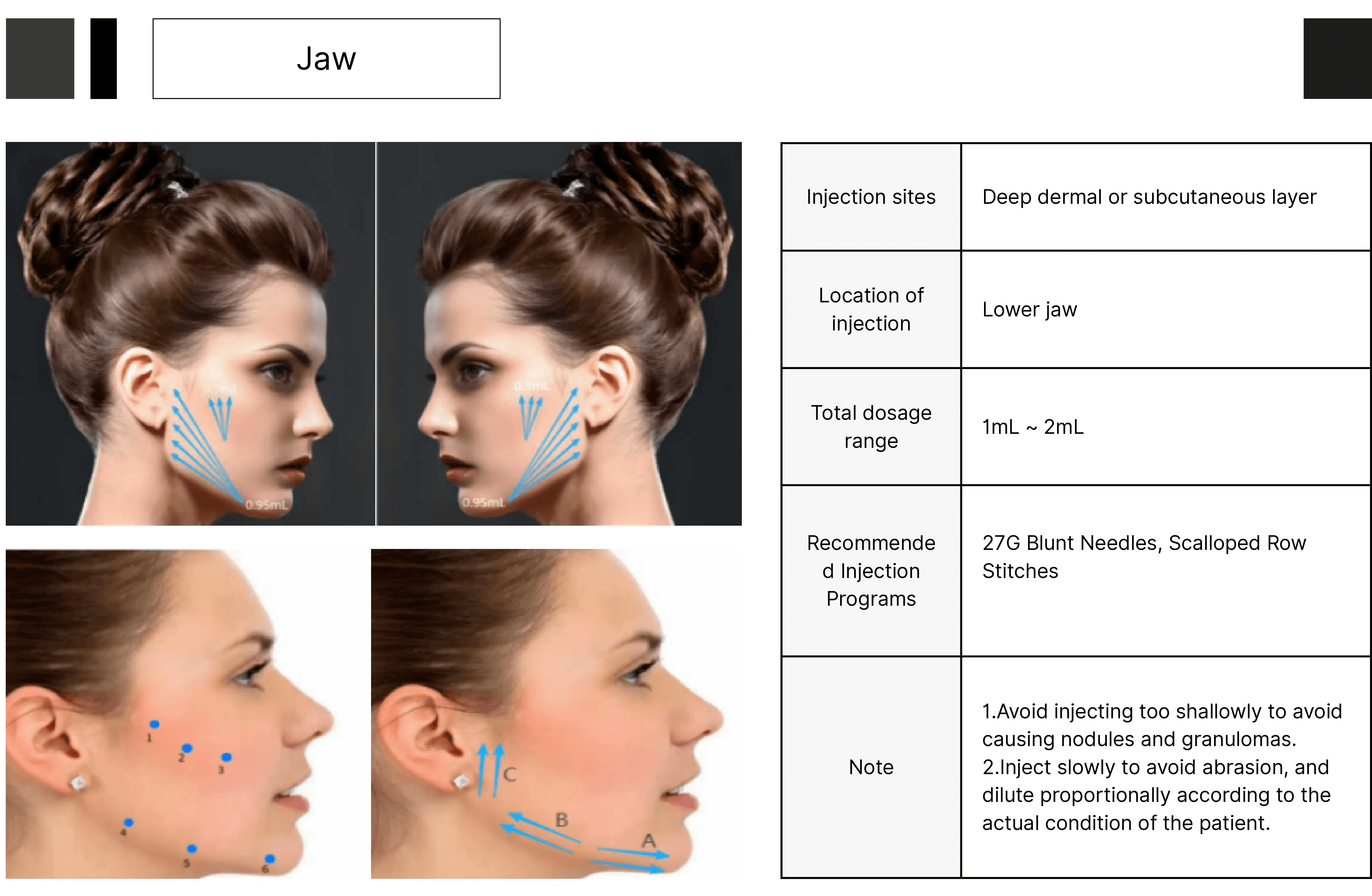
HA-CaHA facial filler gel, suitable for correcting deep facial skin depressions, enhancing cheek fullness, treating wrinkles, marionette lines, and sculpting the jawline, as well as restoring/correcting facial fat loss (atrophy) in AIDS patients. Diluted in varying concentrations, it can be used for wrinkle repair on the face, neck, arms, abdomen, buttocks, and other areas.
6.CaHA Injectable Facial Filler Working Principle
Biodegradable
The degradation process synchronizes with new collagen synthesis: the gel particle matrix degrades first, creating space for new collagen formation; calcium hydroxyapatite microspheres stimulate collagen production before degradation. Degradation products are absorbed and metabolized by the body, ultimately establishing a virtuous cycle of sustained collagen support. This ensures safety while maintaining long-lasting filling effects.
Stable Calcium Hydroxyapatite Microspheres
This biocompatible calcium hydroxyapatite filler, when injected subcutaneously, leaves its hydroxyapatite microspheres permanently at the injection site. These microspheres stimulate collagen regeneration to build a robust autologous collagen scaffold, delivering natural, long-lasting structural support.
Lidocaine for Local Anesthesia
The formulation contains lidocaine, providing short-term local anesthesia to ensure a comfortable injection experience for patients.
Long-Lasting Yet Non-Permanent Results
The core mechanism involves a two-phase action of “immediate filling + long-lasting collagen stimulation” to restore facial volume and enhance facial contours. Sodium hyaluronate gel provides support and hydration before being naturally degraded and metabolized. Hydroxyapatite microspheres act as a biological scaffold, continuously stimulating the production of new autologous collagen to remodel stable tissue structures, achieving natural, long-lasting facial rejuvenation.
7. Usage Guidelines and Safety
Sterilization and Single-Use Precautions
Ensuring Safe Clinical Application
The gel inside the pre-filled syringe is sterilized by high-temperature steam; the accompanying injection needle is sterilized by ethylene oxide.
Comfortable Injection Experience
Lidocaine for Local Anesthesia
The inclusion of 0.3% lidocaine hydrochloride ensures minimal patient discomfort during injection, promoting better tolerance and satisfaction.
Precautions
Strict adherence to sterile surgical procedures is required during use. Refer to the enclosed instructions for details.
Products Status

Mechanism of action
Rveiling® HA-CaHA Composite Filler: Dual-action in one syringe. Deeply builds firm structural support to lift facial contours; finely sculpts superficial layers to create natural shaping power for plump, dimensional skin. Simultaneously stimulates collagen regeneration for long-lasting firmness and skin texture improvement.
Reveiling® HA-CaHA facial filler, when implanted into soft tissue, undergoes gradual degradation where the gel matrix breaks down before the CaHA microspheres. These microspheres provide long-term structural support while stimulating natural collagen regeneration.

Revealing® HA-CaHA Implant Tissue: Pre- and Post-Treatment Effect Comparison Long-lasting activation of collagen regeneration Stabilizes facial tissue volume Reshapes smooth contour lines Achieves a natural, youthful facial appearance.
Back of the hand
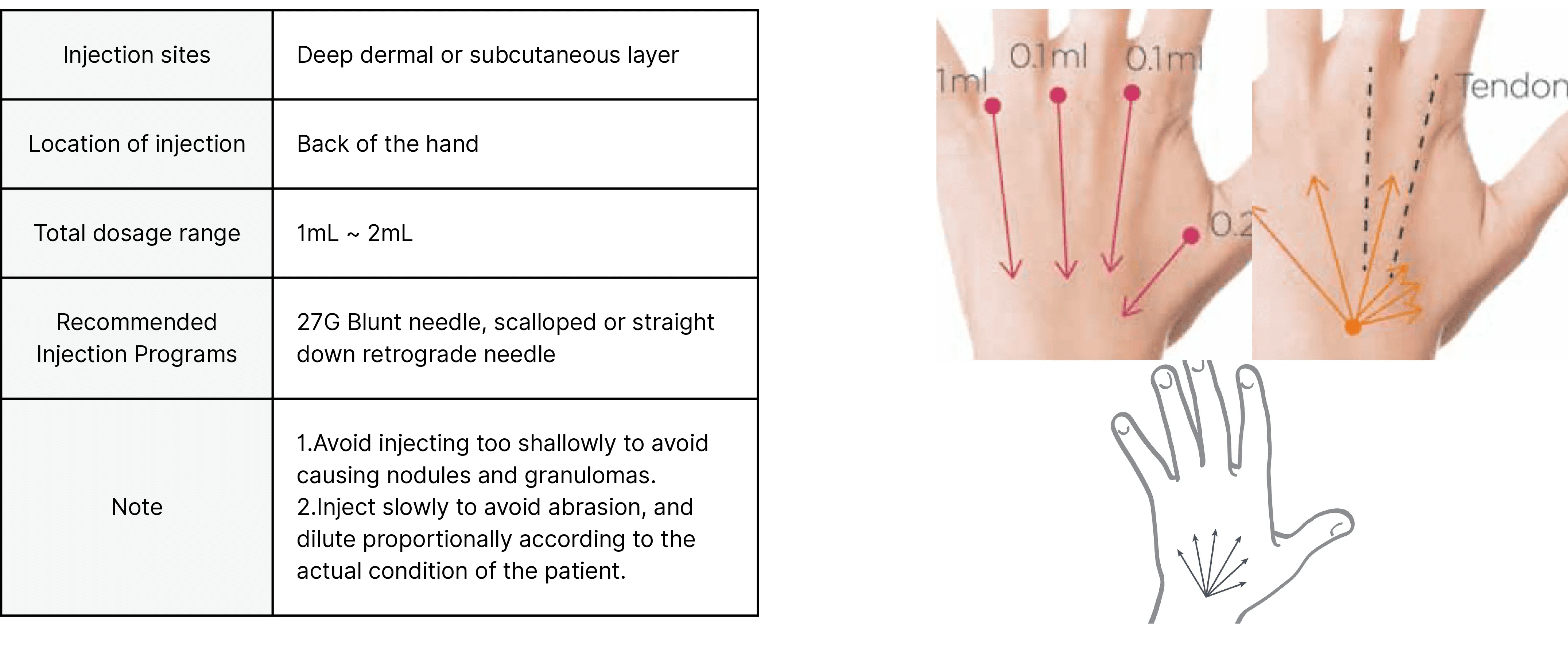
Key attributes
Transport conditions | transported at normal temperature, the transport cycle should not exceed1 month |
|---|---|
Storage condition | 2-25°C,prohibit freezing, avoid sunlight |
Specification | 1 mL/syringe 2 mL/syringe 3 mL/syringe |
Transport Pakeage | 50-100 pieces per box |
HA Gel Carriers | 69.0% |
CaHA Microsphere | 30.7% |
Lidocaine | 0.3% |
Injection Areas |
It should be used by an authorized practitioner. Do not re-sterilize or mix with other products. |
Injection Depth | Subcutaneous tissue, from the middle to deep layers; periosteum, upper layer |
Duration | 12-24 Months |
Before treatment:
Tell your physician about all medications you are taking and your past treatment history.
Foods that increase the chance of bleeding and cause bruising should be avoided for the time being, such as: ginkgo, fish oil, aspirin drugs, vitamin E, etc.
Consult your physician if you are allergic to the drug, breastfeeding, or during pregnancy.
After treatment :
Avoid alcohol and strenuous exercise for twenty-four hours.
Apply light make-up after twenty-four hours and avoid the location of the pinholes.
Intrusive treatments, facial massages, and facial aesthetic procedures are not recommended for two weeks.
Avoid prolonged sun exposure, exposure to UV light, cold temperatures, and hot treatments.
Temporary redness, swelling, soreness and bruising are natural and will subside in five to seven days.
FAQs
1. What is Injectable Hydroxyapatite Microsphere Facial Filler ?
Our calcium hydroxyapatite injectable facial filler is formulated with biocompatible calcium hydroxyapatite microspheres suspended in a biodegradable gel particle carrier. Following implantation, the gel particles gradually degrade and metabolize, while the microspheres provide long-term structural support and stimulate natural collagen regeneration.
2. Can we customize packaging for OEM or private label production?
Yes. As a professional B2B manufacturer and supplier, we offer OEM and ODM customization services, including logo and packaging design. We support clinics, distributors, and medical aesthetic brands with flexible collaboration models.
3. What are the primary applications of injectable calcium hydroxylapatite facial fillers?
This injectable implant filler is suitable for correcting moderate to severe facial wrinkles, contouring, wrinkle reduction, skin folds, and restoring volume loss in the hands. Injected into the deep dermis or upper soft tissue above the periosteum, it delivers non-permanent yet long-lasting enhancement effects.
4. How long do the effects of hydroxyapatite microsphere facial fillers last?
Typically, results persist for 24 to 36 months or longer, depending on the injection site, technique, and patient metabolism. The calcium hydroxyapatite microspheres continue stimulating collagen growth, delivering sustained volume and firmness over time.
5. What certifications and quality standards do you provide for B2B partners?
Our manufacturing strictly adheres to ISO 13485, GMP, and CE certification—globally recognized standards for medical device production quality and safety. Each production batch undergoes exhaustive biological performance testing covering biocompatibility, sterility, pyrogen-free status, and functional integrity. This rigorous process ensures 100% batch compliance, delivering consistent reliability you can trust.
Other Products