Type
Fat-dissolving
Application
Suitable for double chins, abdomen, thighs, and other stubborn fat accumulation areas
Place of Origin
China
Brand Name
Rveiling®
Product Description
Rveiling® Deoxycholic Acid Lipolysis Injection, with the primary ingredient deoxycholic acid, is a naturally occurring free bile acid in the human body and an endogenous substance produced during digestion. It emulsifies and dissolves dietary fats in the intestines, facilitating their breakdown and absorption by lipase. The Deoxycholic Acid Fat-Reducing Injection is a non-surgical, injectable therapy for localized fat reduction. It primarily works by disrupting fat cell membranes, emulsifying fat into fine particles, and subsequently metabolizing and excreting these particles through the lymphatic system. This achieves targeted fat reduction. Clinically validated for localized fat contouring, it is not a substitute for systemic weight loss. Suitable for stubborn fat deposits in areas such as the double chin, abdomen, and thighs.
Product Name
Rveiling® DCA-Containing Lipolysis Injection Solution
Product Ingredients
Water for Injection, Deoxycholic Acid, Sodium Hyaluronate
Product Specifications
5mL per vial, 5 vials per box
Indications
Chin and other areas with localized fat deposits
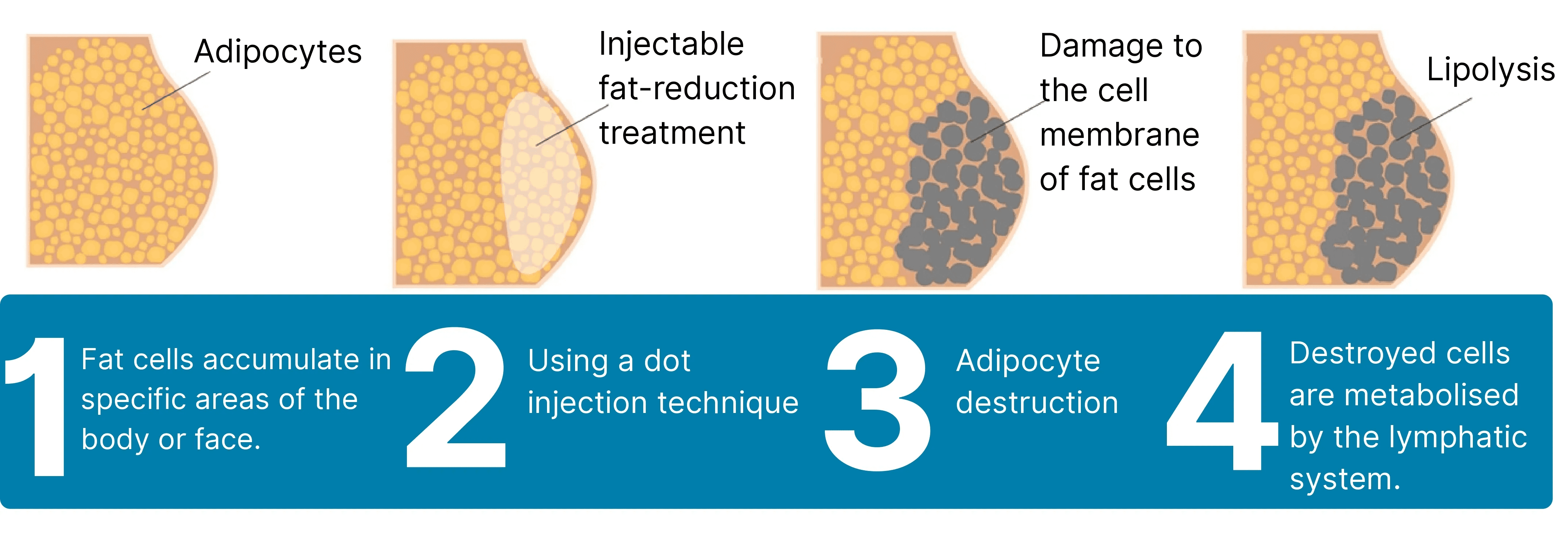
Mechanism of Action
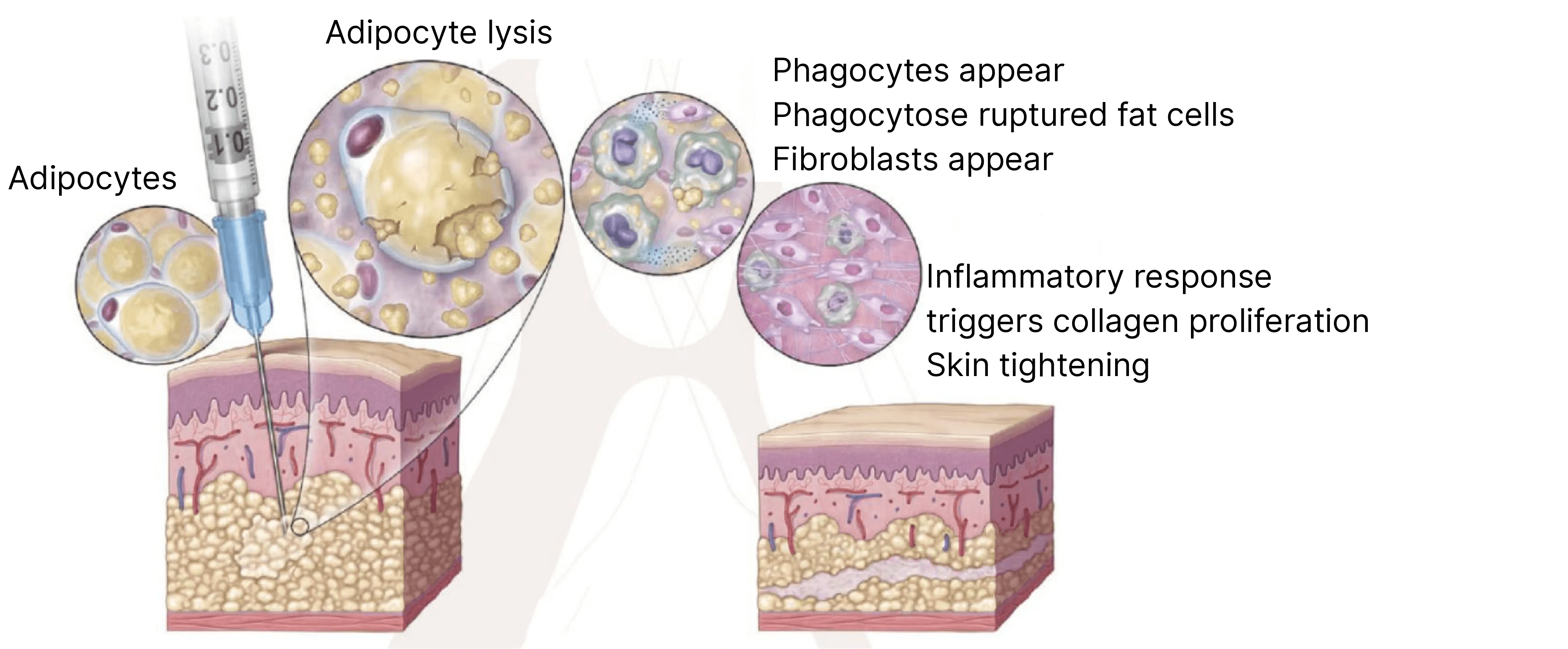
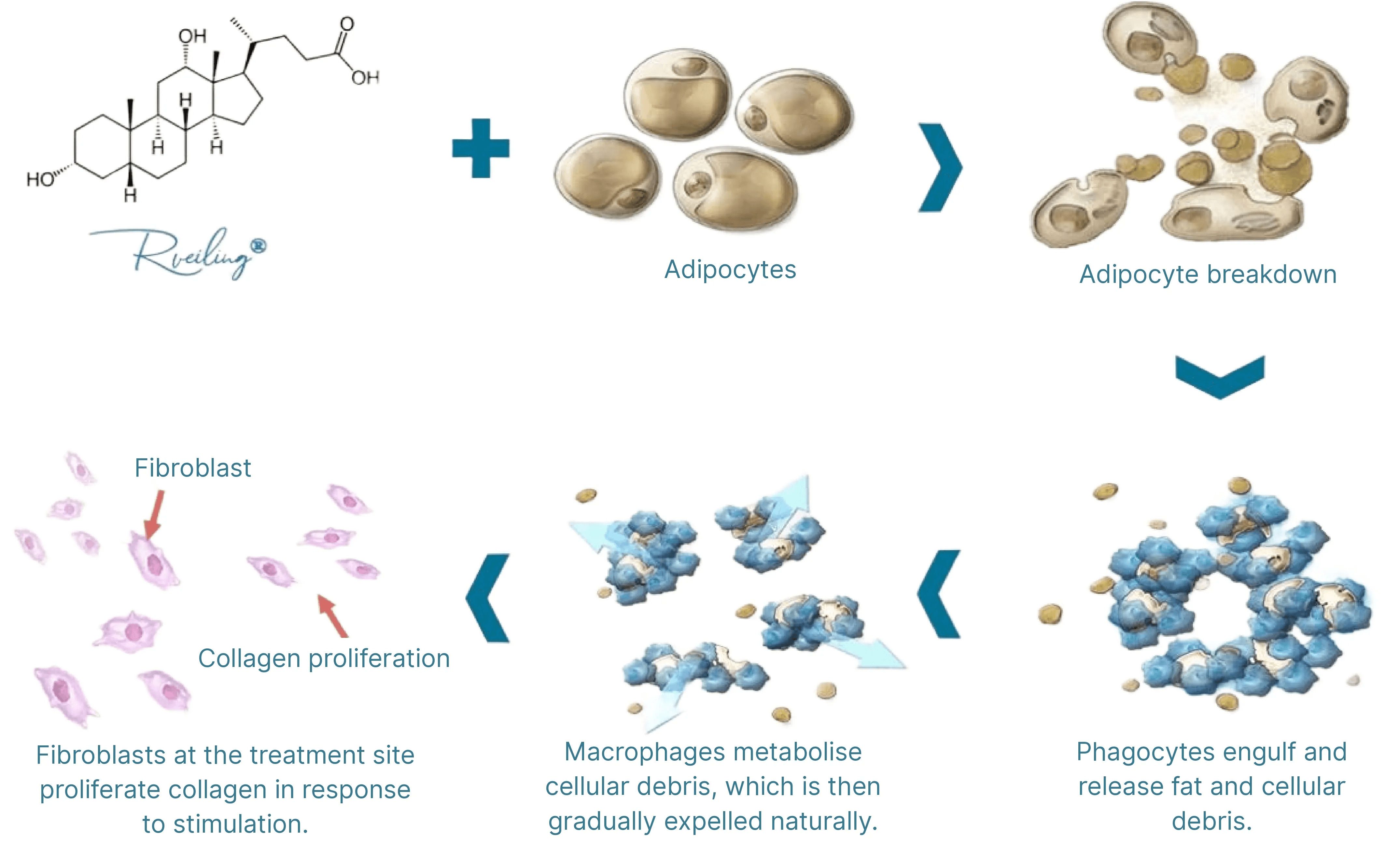
The primary active ingredient, Deoxycholic Acid (DCA), disrupts fat cell membranes, releasing cellular contents. This triggers a localized inflammatory response involving macrophage infiltration and fibroblast aggregation, accelerating fat metabolism. The fat is subsequently eliminated through the lymphatic system as part of normal bodily metabolism, achieving “permanent fat reduction.” This process induces collagen regeneration, promoting skin contraction at the injection site, with results stabilizing approximately 28 days post-treatment. The deoxycholic acid in the injection solution is a synthetic compound, eliminating risks of infection from human or animal-derived pathogens. It fully degrades and exits the body within 24 hours post-injection, restoring endogenous deoxycholic acid levels to pre-injection status, ensuring excellent safety. This ingredient is clinically validated for localized fat contouring but cannot replace systemic weight loss.
Applications
Fat-dissolving injections, facial contouring. Results typically last 3-5 years or longer after 2-4 treatment sessions.
Suitable Candidates
Individuals with localized fat deposits (e.g., double chin, submental area, upper arms, abdomen, thighs).Those with good skin elasticity and no significant laxity.
Shelf Life: 18 months
Storage Conditions: Store at 2–25°C. Do not freeze.
Transportation Conditions: Room temperature (≤30°C) transport must not exceed 1 month; otherwise, cold chain transportation is required.
Note: This product is sterile. Strict adherence to sterile surgical procedures is mandatory during use.
Usage Details: Refer to the package insert.
Key Highlights
Targeted Fat Reduction: Destroys and removes localized fat cells in areas such as the double chin, abdomen, and thighs.
Active Ingredient – Deoxycholic Acid: The core component damages the fat cell membrane, emulsifies fat, and breaks it down into fine particles.
Natural Metabolism: The broken-down fat is metabolized and excreted through the lymphatic system.
Clinically Proven: Demonstrated efficacy and safety for localized fat contouring, though not intended for systemic weight loss.
Minimally Invasive: Offers a non-surgical alternative for facial and body sculpting with minimal downtime.
Scientific Dissolution, Precise Metabolism
● High-Efficiency Emulsification: Targeting the cell membranes of stubborn fat cells, it emulsifies and decomposes them.
● Accelerate Metabolism: Promote the excretion of decomposed fat from the body through the body's natural metabolic system.
● Firming the Skin: The synergistic ingredients help stimulate collagen, assisting the skin in restoring firmness and elasticity while reducing fat.
Key attributes
Deoxycholic Acid | 10mg/ml |
|---|---|
Sodium Hyaluronate | 3mg/ml |
Applicable Crowd | For improving moderate to severe contour prominence or localized fat accumulation causing facial fullness in adults, such as double chin, arms, abdomen, thighs, etc. Suitable for individuals with good skin elasticity and no significant sagging. |
Contraindications | Pregnant women, breastfeeding women, and women during menstruation. Individuals with heart disease, diabetes, or immune system disorders. Those allergic to soy, eggs, or any medication ingredients. |
Storage Condition | 2–25°C. Do not freeze. |
Recommended Treatment Course | 3 to 5 sessions constitute one course, with sessions spaced 15 to 20 days apart (the number of courses may be appropriately increased based on skin condition). |
Injection Depth | Treatment plans should be tailored to individual differences. A professional physician should assess skin thickness and fat characteristics to determine the appropriate injection depth for each area. |
Specific Details | Refer to the package insert for detailed instructions. |
FAQs
1. What is Deoxycholic Acid (DCA) Fat Dissolving Injection used for?
Deoxycholic Acid (DCA) injection is a cytolytic injectable used to improve the appearance of moderate to severe submental fullness, commonly called a “double chin,” in adults. Public U.S. drug information explains that it works by destroying fat cells in the treated area. Just as important, current official and reference sources also state that the safe and effective use of deoxycholic acid outside the submental area has not been established. For a factory or OEM/ODM product page, the most professional positioning is to describe DCA as a regulated injectable for submental fat appearance improvement, not as a broad body-fat solution for any area.
2. How does Deoxycholic Acid injection work?
Deoxycholic acid is described in official public references as a cytolytic medication that works by breaking down fat cells in the treated fatty tissue. Once those cells are destroyed, the body gradually clears the cellular debris over time. In practical product-page language, this means DCA is not marketed as a temporary “slimming” cosmetic in the same way as a massage serum or topical contour product. It is an injectable active with a specific mechanism of action and a procedure-based treatment context. Because of that, the manufacturing, labeling, and claims strategy should reflect medical or aesthetic-clinic use standards rather than ordinary skincare positioning.
3. Is Deoxycholic Acid injection safe for all treatment areas?
No. Current official U.S. HCP information states that the safe and effective use of deoxycholic acid for subcutaneous fat outside the submental region has not been established and is not recommended. This is one of the most important points for a product information page, because many lower-quality product pages overgeneralize DCA as a universal fat-dissolving injection. A more credible and compliant answer is that market-specific approval, intended use, and labeling must be confirmed before promoting any treatment area claims. In addition, deoxycholic acid injection is contraindicated in the presence of infection at the injection sites, according to current labeling.
4. What are the common side effects and risks of DCA fat dissolving injections?
Public medical and official drug information consistently note that common reactions can include swelling, bruising, pain, numbness, redness, firmness, and tenderness at the injection site. More serious warnings in current clinical references include trouble swallowing, facial muscle weakness, uneven smile, nerve injury in the jaw area, skin ulceration, and injection-site complications that require prompt medical attention. This means a professional factory page should avoid casual phrases such as “no downtime” or “no risk.” A better approach is to state clearly that DCA injections must be used by qualified healthcare professionals, and that safety depends on proper patient selection, technique, and market-compliant instructions for use.
5. What should buyers check when choosing a Deoxycholic Acid injection factory or bulk supplier?
For B2B buyers, the most important checks are regulatory status, manufacturing standards, formulation specifications, sterility controls, packaging format, market labeling support, and documentation readiness. Current FDA labeling describes KYBELLA as a sterile, colorless solution for subcutaneous use at 10 mg/mL, supplied in single-patient-use vials, which shows how tightly defined injectable specifications can be in a regulated market. A strong supplier page should therefore focus on whether the factory can support GMP-level manufacturing expectations, sterile filling capability, batch consistency, COA and technical documentation, and market-specific compliance requirements. Because DCA is not a standard cosmetic ingredient in many jurisdictions, buyers should evaluate it as a regulated injectable product, not as a routine beauty serum.
Other Products












